‘Shifting goalposts’: Coquitlam lab stuck in bureaucratic quagmire
Omega Laboratories has been waiting to get a COVID antibody test approved for nearly 9 months

Midway through the COVID-19 pandemic, long wait times on PCR test results were holding up film productions in Hollywood North.
It was one U.S.-based company’s ticket into the Canadian outpatient lab market.
“Time is money,” said Dr. Gio Miletto, medical director of the Omega Laboratories’ Coquitlam facility. “(Studios) couldn’t get the services they needed, essentially.”
Local news that matters to you
No one covers the Tri-Cities like we do. But we need your help to keep our community journalism sustainable.
But two years after being “begged” by a major movie studio to set up shop in the Lower Mainland, Miletto said the company is on the verge of shutting down business in B.C.
They have been waiting to get a single test approved for nearly nine months by the industry’s accreditation body.
“This is not normal,” Miletto said. “This is not how it works in any other jurisdiction that Omega has experience in.”
Omega’s executives have been vexed by what they claim is an overbearing licensing process that deviates from any standard they’ve experienced.
The Diagnostic Accreditation Program (DAP), under the College of Physicians and Surgeons of BC (CPSBC), regulates and controls market access for the entire B.C. lab industry.
“We’ve never run into an organization like this,” said CEO Bill Corl, noting Omega has built labs in many countries around the world.
Omega is accredited under the International Organization for Standardization (ISO), the same standards regime that DAP uses. However, B.C. labs are required to acquire licensing locally through the CPSBC.
Since starting operations in Coquitlam in November 2020, the company has only managed to get one PCR test approved.
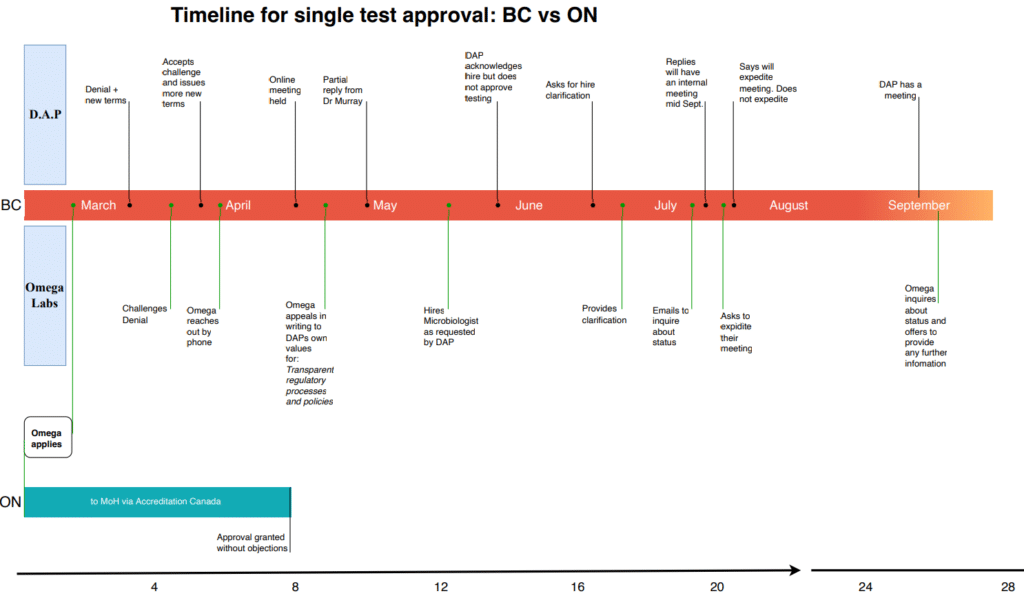
Omega has been trying to get a COVID antibody test approved since March, 2022.
The company opened a second Canadian lab in Toronto in July 2021, which has 21 tests approved across 18 drug classes. The same antibody test took eight weeks to get approved.
“We’re talking about the same Health Canada equipment, same operating procedures, same level of expertise,” Miletto said. “There is global guidance on the use of antibody tests. This is not some kind of mystery test.”
He claims the discrepancy between the two province’s accreditation process has served as a controlled experiment showing something is amiss with B.C.’s regulatory system.
After investing over $2 million into the facility, Omega has had to cut back hours for its 20 employees.
Miletto described the economic situation as “dire.”

B.C.’s lab industry
LifeLabs has dominated the province’s lab industry since buying out its main competitor, BC Biomedical, in 2013.
The company owns approximately 90 percent of the private outpatient labs in B.C., and holds a lucrative public service contract with the provincial government, enabling it to bill the Medical Services Plan more than $263 million last year alone.
There has been a provincial moratorium on the issuance of new public service contracts since 2015, so legally, Omega can only bill uninsured clients, such as companies in the film or travel industries.
Miletto said the moratorium has effectively given LifeLabs a provincially mandated monopoly on the industry, noting that in Ontario, there is an open bidding process for service provision.
The Canadian outpatient lab industry has become an increasingly concentrated both in terms of specimen collection and analysis, according to Andrew Longhurst, research associate with the Canadian Centre for Policy Alternatives.
He said B.C. was caught “a bit flat footed” when it came to testing capacity at the outset of the pandemic.
Many for-profit players in typically engage in what is often referred to as “cream skimming,” according to Longhurst.
The term is borrowed from the insurance industry, and refers to taking the easiest, most-lucrative, high-volume tests out of the public sector’s hands and charging governments for service. The more expensive, labor-intensive testing is left to public labs.
“One of the big issues, and this is really quite evident with the pandemic and where the money is … is the markups on all of the COVID testing and all the private pay lab work are significant,” Longhurst said. “Without an assessment, what kind of value do we get when we have one big, very significant player?”
He said there is a strong argument that all B.C.’s testing services would be cheaper over the long run if they were publicly delivered.
‘Shifting goalposts’
Film productions often require co-ordination between hundreds of moving parts and people on tight deadlines.
In early 2021, studios were waiting up to four days to get union-mandated results back, according to Miletto.
He said they wanted to test employees in the morning to have them on set later that day, and Omega’s typical turnaround time is four hours.
The local buzz around the company led to it being featured in CBC and Global News reports.
One radio station even titled an interview segment with Miletto as “LifeLabs monopoly could soon be ended.”
But Omega executives claim their trouble with DAP began almost immediately after opening its B.C. lab.
The company had received accreditation for its PCR test in November, but wasn’t able to service their clients until January, according to Corl.
“(Studios) started to send samples to us, and all of a sudden DAP contacted us and told us we had to stop,” he said.
Corl claims they weren’t allowed to resume testing until legal representatives from a major studio complained to the Ministry of Health (MOH), stating they were close to shutting down productions. The MOH would not confirm this meeting.
Miletto said lab assessment standards are typically objective, such as keeping a freezer below a certain temperature, but the DAP inspectors seem to interpret new rules “on the fly.”
“This isn’t some standard that we missed,” Miletto said. “Everything keeps shifting, it’s death by 1,000 cuts.”
In one case, DAP overruled Coquitlam Fire Department inspectors; in another, they required further validation of recently purchased Health Canada-approved equipment.
Omega claims these are not normal practices, while the CPSBC maintains they are industry standards.
Since starting operations in B.C., Omega said they’ve flagged for 105 major deficiencies, with DAP sometimes threatening to shut down the lab in a matter of days.
By contrast, Omega’s Toronto lab has only been issued 22 minor recommendations.
When the company applied for accreditation for a COVID antibody test, DAP denied their application outright.
The rejection letter stated the committee had concluded there was “little clinical utility” for an antibody test.
The letter also stated no facility would be approved without confirmation from the BC Centre for Disease Control (BCCDC) that their test was in support of the public health effort.
Miletto said he was shocked by the response.
“We’re still in a pandemic, we’ve got demand from a non MSP client, and the test is already being provided by LifeLabs,” he said. “It doesn’t make any sense.”
Miletto’s appeal pointed out the BCCDC, the U.S., and European Union CDCs, all plainly describe the test’s clinical utility on their websites.
DAP ultimately granted their appeal, but with a new list of requirements. Milletto described it as “shifting goalposts.”
The requirements were met by mid-May, but the company says it has still not received approval.
Miletto said he does not disagree with the need to heavily regulate the lab industry, but this is “above and beyond” what should be required.
“No one can run a business like this,” he said.
Other labs’ perspectives
Omega is not the only lab with complaints about DAP.
The Dispatch spoke to one CEO running a small specialized lab in B.C. testing samples from clients across North America. He requested anonymity over concerns related to future licensing.
The CEO said DAP is more restrictive than any U.S. regulator, adding that it took them more than a year to get a test approved.
He said even among the most rigorous U.S. organizations, such as New York State’s, labs are allowed more flexibility, and can continue operating while they address citations.
In contrast, the CEO claimed DAP threatens to revoke accreditation if minor issues can’t be fixed by a set date, according to the CEO.
“When we look at the things that we are failing on, it’s not going to affect test results,” he said. “It’s above and beyond patient care, I think that’s what we get frustrated about.”
Small labs are treated no differently than large organizations with far more resources, the CEO said. He added the regulators often lack expertise on certain types of tests.
Some of these complaints were echoed by Dr. Hans Frykman, medical director of UBC’s neuroimmunology lab. He also runs three other private labs in B.C.
Frykman said he has less staff than larger labs, and DAP regulators do not always have the right competency to assess complex forms of testing.
Larger U.S. accreditation bodies like the College of American Pathologists and the Clinical Laboratory Improvement Amendments are more efficient and knowledgeable, but not always as detail oriented, according to Frykman.
“DAP is very meticulous,” he said. “We also argue with them about things that could be called ridiculous … They can, at times, be pretty harsh.”
But Frykman qualified that there are many different viewpoints on DAP, and the organization has been improving.
He said he prefers how demanding the process is, because in his opinion, some smaller labs should not be operating.
Frykman said he was concerned over the number of small “sketchy” labs trying to take advantage of PCR testing through the pandemic, sometimes illegally.
“We take quality very, very seriously.”
Transparency
One of Omega’s main complaints is not understanding who on the DAP committee is making decisions regarding their accreditation.
Miletto said B.C. ‘s process is opaque in comparison to other jurisdictions. He notes that because CPSBC is self regulating they have little recourse through the province.
The company appealed to Coquitlam city council in late-October, local MLA Selina Robinson, and various provincial ministers to ask for assistance to expedite their approval process.
Robinson said she has advised the company to file an official complaint with the CPSBC.
The company’s uncertainty is not eased by the DAP committee being chaired by the director of LifeLabs, their main competitor.
The provincial government introduced the new Health Professions and Occupations Act (HOPA) on Oct. 19, 2022, which will replace the regulatory system governing B.C.’s health colleges.
While HOPA will not affect DAP’s accreditation procedures, it does build on several provisions relating to board member’s conflicts of interest, including adding an actual definition.
The CPSBC told the Dispatch via email they said they cannot comment on individual cases, and declined an interview request.
Responding to general questions by email, a DAP spokesperson told the Dispatch that committee members have to declare conflicts of interest on an annual basis, and abstain from accreditation decisions when there are conflicts.
“. . . all processes have been assessed and found to meet all appropriate international requirements for impartiality and conflict of interest,” the spokesperson wrote.
They stated that DAP “maintains a consistent process for all private and public facilities.”
Asked about the speed of the accreditation process in B.C. compared to other provinces, a DAP spokesperson responded that the agency “does not have access” to that information.
There is an inherent difference between facilities, the spokesperson explained. That difference “limits the effectiveness of tracking the time it takes to obtain accreditation.”
In terms of weighing in on the clinical utility of a specific test, the spokesperson explained that the COVID-19 pandemic was the rare example of a time when the agency deviated from its standard process.
The DAP eventually provided accreditation for all COVID-19 services, “for all facilities with a medical director who have the appropriate credentials to provide COVID serology testing services.”